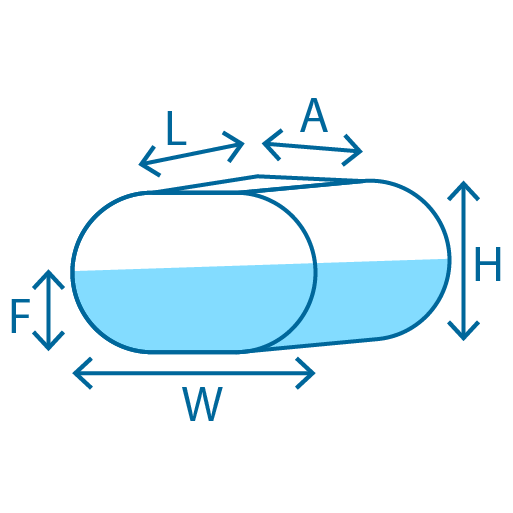
The width of the tank is A, the height of the tank is B, and the height of the elliptical segment h corresponds to the fluid surface. For this calculator to give accurate results the geometry of the tank must be entered. This shows the percentage of tank capacity that is filled. The calculator above can tell you volume of the fluid in the tank, or produce a dip chart for future reference. The volume of the liquid contents held in the tank are displayed here in your preferred volumetric units. The amount of liquid in the tank is calculated here. If you only know the outside diameter of the tank, it is okay to use it, but the error of the calculated volume will be dependent on the thickness of the tank wall. If you only know the radius of the tank, then multiply this by 2 to determine the diameter. Diameter of TankĮnter the inside diameter of the tank, which if you were standing inside the tank would be the widest distance from one side of the tank to the other. If you only know the outside height of the tank from top to bottom, then the accuracy of the volume calculated will depend on the positional height of the overflow outlet at the top of the tank and the feed line at the bottom of the tank. Specify the following reading and tank dimensions Height of Liquid in TankĮnter the height measured from the minimum level to the top of fluid in the tank Height of TankĮnter the height of the tank which should be the difference in height between when the tank is empty and when it is full. So, for a household of 4 people with an estimated liquid capacity of 100 liters per person, you would need a septic tank of approximately 800 liters. Tank Volume: 400 liters/day × 2 days 800 liters. Use this calculator tool to determine the amount of contents in a upright cylinder shaped liquid storage tank from the measured height of fluid in the tank. Daily Flow: 4 people × 100 liters/person 400 liters/day.
0 Comments
One neurotransmitter that has been very extensively studied is dopamine, C 8H 11NO 2. This process is called exocytosis (see Figure 6.6). These neurotransmitter molecules are stored in small intracellular structures called vesicles that fuse to the cell membrane and then break open to release their contents when the neuron is appropriately stimulated. Chemical signaling occurs at the interface between different neurons when one of the cells releases molecules (called neurotransmitters) that diffuse across the small gap between the cells (called the synapse) and bind to the surface of the other cell. Specialized cells called neurons transmit information between different parts of the central nervous system by way of electrical and chemical signals. 
(b) Information is transmitted in brain tissue and throughout the central nervous system by specialized cells called neurons (micrograph shows cells at 1600× magnification). (a) A typical human brain weighs about 1.5 kg and occupies a volume of roughly 1.1 L. The molar mass of an element (or compound) is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol) (see Figure 6.1). The masses of 1 mole of different elements, however, are different, since the masses of the individual atoms are drastically different. This constant is properly reported with an explicit unit of “per mole,” a conveniently rounded version being 6.022 ×10 23/mol.Ĭonsistent with its definition as an amount unit, 1 mole of any element contains the same number of atoms as 1 mole of any other element. This large number is a fundamental constant known as Avogadro's number (N A) or the Avogadro constant in honor of Italian scientist Amedeo Avogadro. A mole of substance is that amount in which there are 6.02214076 × 10 23 discrete entities (atoms or molecules). The mole provides a link between an easily measured macroscopic property, bulk mass, and an extremely important fundamental property, number of atoms, molecules, and so forth. One Latin connotation for the word “mole” is “large mass” or “bulk,” which is consistent with its use as the name for this unit. It provides a specific measure of the number of atoms or molecules in a sample of matter. The mole is an amount unit similar to familiar units like pair, dozen, gross, etc. This experimental approach required the introduction of a new unit for amount of substances, the mole, which remains indispensable in modern chemical science. Today, we possess sophisticated instruments that allow the direct measurement of these defining microscopic traits however, the same traits were originally derived from the measurement of macroscopic properties (the masses and volumes of bulk quantities of matter) using relatively simple tools (balances and volumetric glassware). However, because a hydrogen peroxide molecule contains two oxygen atoms, as opposed to the water molecule, which has only one, the two substances exhibit very different properties. For example, water, H 2O, and hydrogen peroxide, H 2O 2, are alike in that their respective molecules are composed of hydrogen and oxygen atoms. 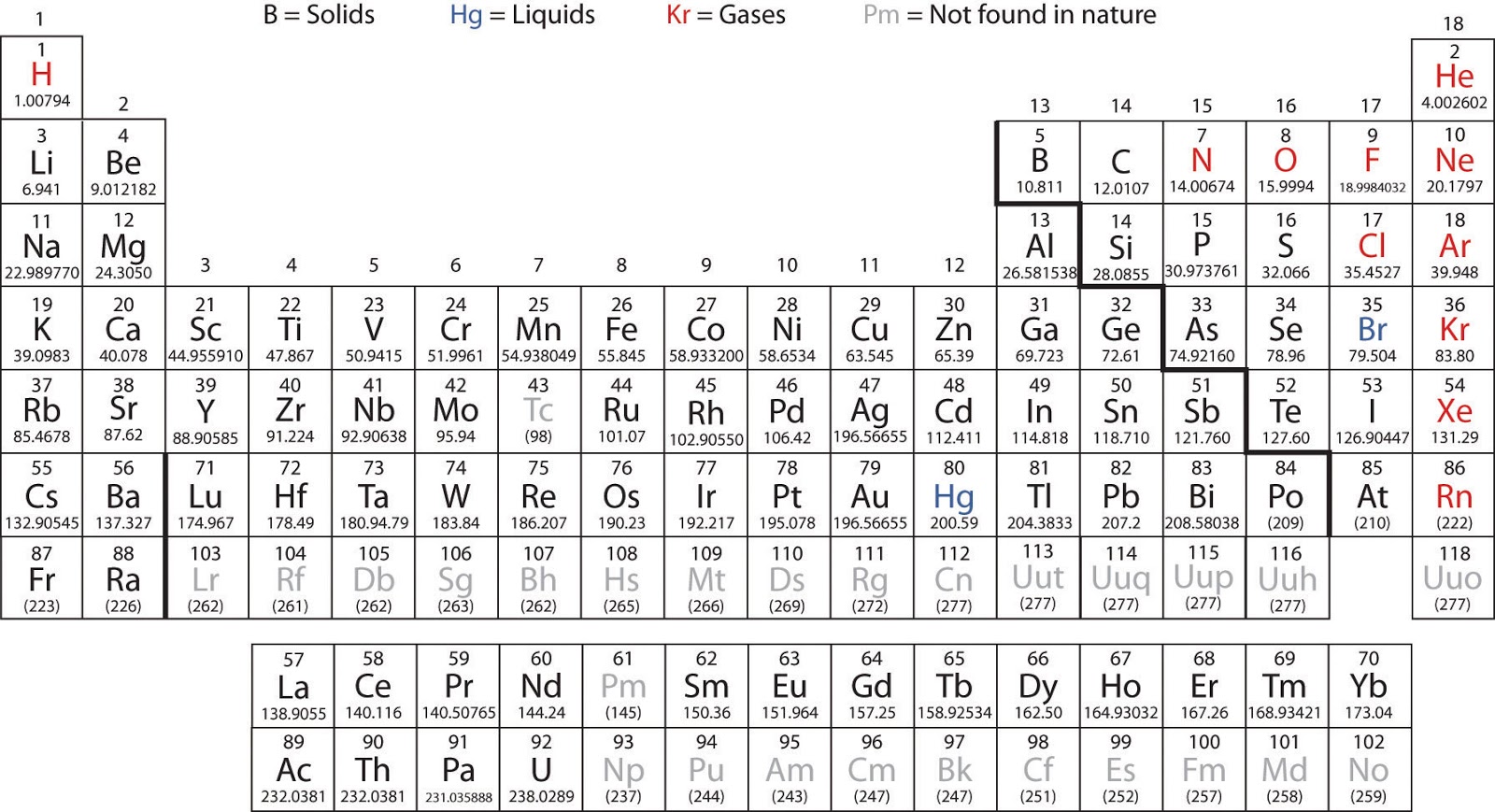
The identity of a substance is defined not only by the types of atoms or ions it contains, but by the quantity of each type of atom or ion. :max_bytes(150000):strip_icc()/PeriodicTable-56a12c983df78cf772682271.png)
View available translations of the content. Download the content in PDF, Microsoft Word, or other format. Questions and Tasks Add a note to the content. Kinetic Molecular Theory, Real GasesĬontent Foreword Atoms Introduction Matter Measurement The Atom Periodic Table Moles & Mass Light Blackbody Radiation, Photoelectric Effect Atomic Spectra, Bohr Model Orbitals & Quantum Numbers Electron Configurations Periodic Trends Molecules Bonding Nomenclature Lewis Structures Part 1 Lewis Structures Part 2 Molecular Shape Polarity Organic Molecules Isomers Valence Bond Theory Molecular Orbital Theory Interactions Pressure & Gas Laws Combined & Ideal Gas Laws Dalton's Law, Graham's Law, Henry's Law Kinetic Molecular Theory, Real Gases Intermolecular Forces Properties of Water Applications of IMF Phase Diagrams Reactions Stoichiometry Limiting Reactants, % Yield % Composition, Empirical Formulas Energy, Heat, and Work Calorimetry Part 1 Calorimetry Part 2 Enthalpy Part 1 Enthalpy Part 2 Entropy Gibb's Free Energy Climate and Human Impacts Search within this publication Search the entire site Blackbody Radiation, Photoelectric Effect 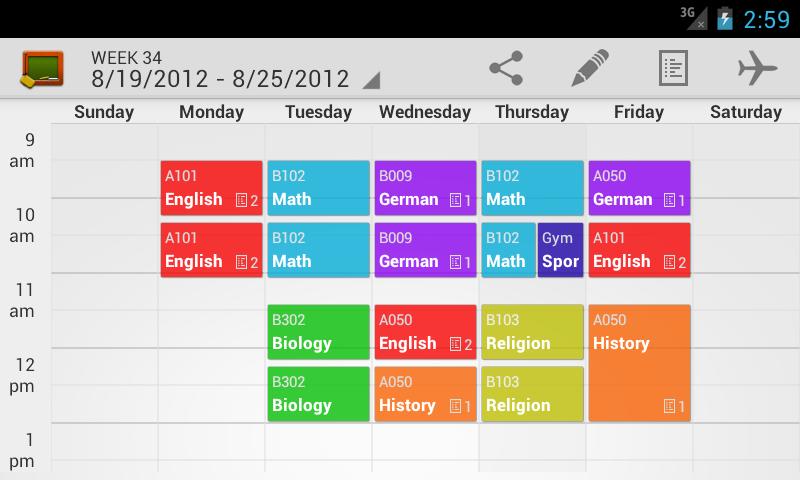
Off Campus means class is held at another location, for example, a local high school.Online Program means it is an online course for students in a 100% online degree program only.Internet means it is an online course for students not in a 100% online degree program.The different campuses include Main, Downtown, Internet, Off Campus, Out-of-State, and Online Programs.If the instructor is Staff, that means an instructor has not yet been assigned to this course. IF NONE OF THE DAYS ARE BLUE, that means the course is asynchronous or is individualized instruction like Independent Study, Master’s Thesis, Doctoral Dissertation, etc. Click on the course title to view more information about a specific section including the course description and pre-requisites.ĭays in blue are the days and times when the class meets. Information related to the course, including Course Subject, Course Number, Section Number, CRN, Title and credit hours. If you are looking for upper-division courses only, refine your search by selecting 3000 – 4999 in the Course Number Range. Search for courses using “ low-cost” (total required learning material cost of $40 or less) or “ free textbooks” (total required learning material cost of $0) which is part of Texas Senate Bill 810 2017 and TEC § 51.542.Honors College Courses are undergraduate-level courses for Honors College students.Upper-Division Courses are undergraduate-level courses with course numbers starting with “3” and “4”. 
Lower-Division Courses are undergraduate-level courses with course numbers starting with “1” or “2”.If you are looking for courses with certain features such as free textbooks, online (Internet), graduate-level, etc., select the appropriate Attribute. Refine your search by selecting only undergraduate, master, or doctoral level courses. Courses exclusively for students in fully online degree programs have a campus of “ Online Programs“.Courses that are online have a campus of “ Internet” ( currently, to search for online courses, you must select “internet campus only” checkbox in the campus section on the class schedule.).Face-toFace courses have a campus of “Main Campus” or “DownTown Campus”.You can use this to filter for courses offered by campus. You can use this to view courses offered during a specific part of term for the semester. Narrow your search by entering the Course Number, Title or keywords related to the course. The term will be displayed as a reminder.īegin entering the subject you want to search. After selecting a term, you’ll see this “BrowseĬlasses” screen where you can enter your searchĬriteria. Students will need to install the customized LockDown Browser for each platform in order to take the exams that require the use of the LockDown Browser. Both platforms have their own customized versions of the LockDown Browser. Respondus Monitor is NOT integrated with WebAssign and McGraw-Hill Connect. How to set up your Coursera faculty or staff account How to set up your Coursera student account Taking a quiz/exam using LockDown Browser in Pearson MyLab Taking a quiz using Respondus in WebCampus Respondus LockDown Browser and Monitor student resources Live proctoring using Respondus LockDown Browser in Zoom Using LockDown Browser with WebAssign and McGraw-Hill Connect Using Respondus Monitor with Pearson MyLab Using LockDown Browser and Respondus Monitor for 3rd party applications Other Respondus training and support resources Setting up a quiz using Respondus Monitor 
Setting up a quiz using Respondus LockDown Browser Respondus LockDown Browser and Monitor faculty resources PointSolutions account registration, WebCampus connection and subscriptionĬreating a Turnitin assignment using Plagiarism ReviewĬonverting an existing Turnitin assignment to use the Plagiarism Review tool Import questions from Word to create a Question List Managing session data and running reports Using the PointSolutions desktop app in the classroom Recommended Zoom screen share default settings Recommended Zoom meeting settings for classes  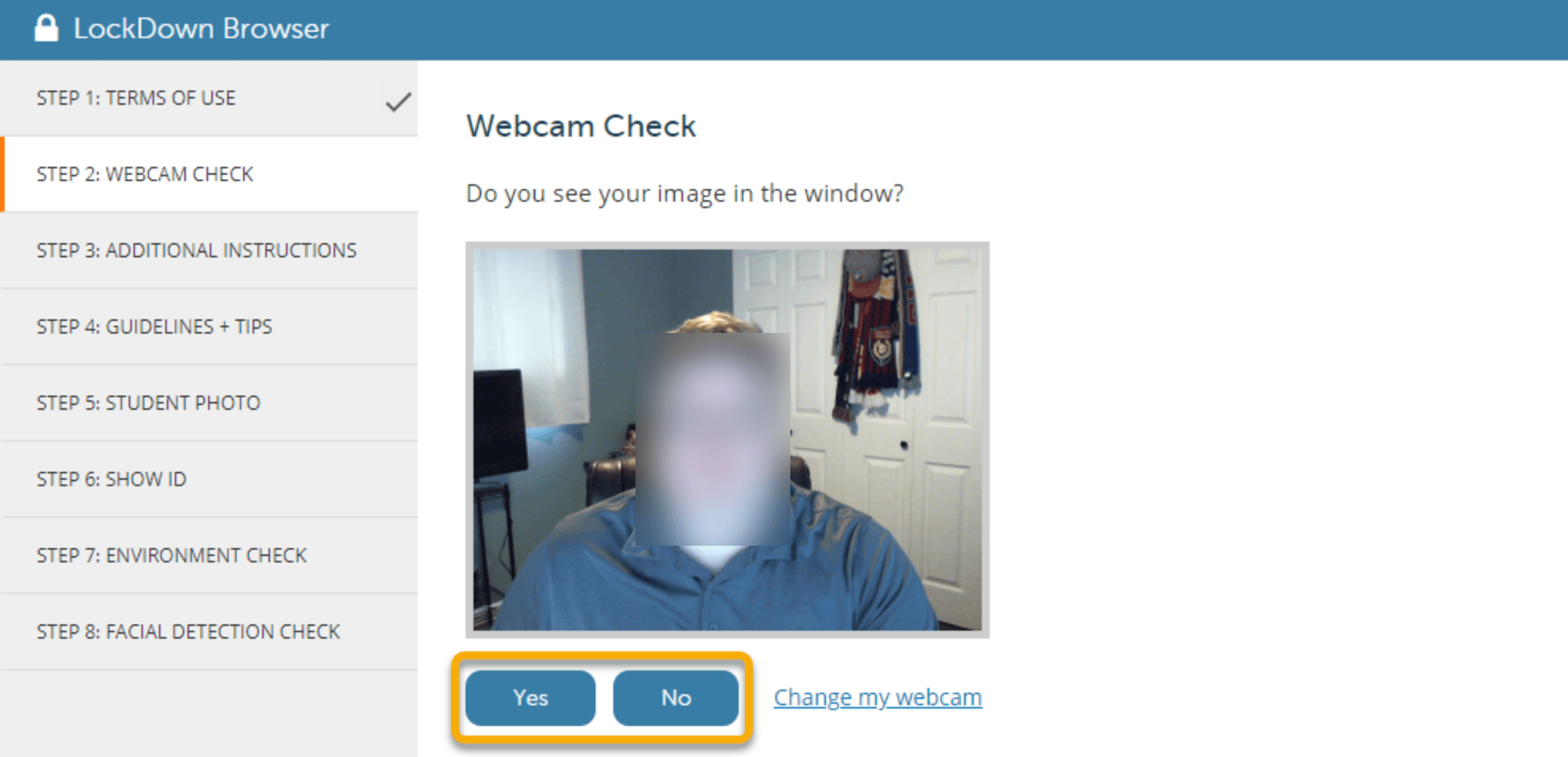
Signing into your University of Nevada, Reno Zoom account Learn how to use the Express Capture application to record your webcam and microphone directly in WebCampus without needing to install additional software.Īdding video from My Media to a course moduleĪdding video from My Media to a content page Recording webcam and mic with Express Capture Sharing videos from My Media with your class via the Media Gallery How to create a video quiz using My Media (Kaltura Video Quizzes) How do I erase my iPad before returning it to university? Faculty iPads through the Digital Wolf Pack Initiativeĭigital Wolf Pack Initiative faculty sessions |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
